|
Therefore, the empirical formula of aluminium oxide is Al 2O 3. Each oxide has a charge of 2-, which means that we can get 6 negative charges by having 3 oxides. Each aluminium has a charge of 3+, which means that we can get 6 positive charges by having 2 aluminiums. Check Your Learning Using the periodic table, predict whether. Group 6 and 7 elements are non-metals and they receive/ gain electrons resulting in them becoming negative ions (they become anions). Lithium (group 1) is a metal, and carbonate is a polyatomic ion Li 2 CO 3 is predicted to be ionic. This means that we can balance the charge of this ionic compound by having 6 positive charges and 6 negative charges. When atoms of nonmetal elements form ions, they generally gain enough electrons to give them the same number of electrons as an atom of the next noble gas in the periodic table. The first number that 3 and 2 go into is 6 (the LCM is 6). The balancing for aluminium oxide is slightly different because the positive and negative charges do not go into each other the 3 positive charge from aluminium and the 2 negative charge from oxygen do not go into each other. Cobalt compounds are used to color glass, ceramics, paints, and cosmetics. Alloys produced with cobalt metal are used in the manufacture of aircraft engines, magnets, grinding and cutting tools, and medical devices and prosthetics. You must know charges on ions formed by Group A main elements. The overall charge for an ionic compound must be 0, which means that the charges from aluminium and oxygen must balance out. What is cobalt Cobalt (Co) is a naturally occurring element found in rocks, soils, water, and air. Carbonate, CO32-, Carbonic acid, H2CO3 (unstable, decomposes into H2O and CO2). Group VIA elements gain two electrons upon ionization, obtaining -2 charges and reaching the electron configurations of the noble gases that follow them in the. 
Oxygen is in group 6 in the periodic table, which means that it will gain 2 electrons resulting in it having a 2 negative charge (O 2-). Name and formula of the acid containing the anion. For a cation, simply use the name of the element and add the word ion (or if you want to be more specific, add cation) after the element’s name.So Na + is the sodium ion Ca 2+ is the calcium ion. Aluminium is a metal and is in group 3 in the periodic table, which means that it will lose 3 electrons resulting in it having a 3 positive charge (Al 3+). Thus, the formula for this ionic compound is Fe 2O 3. To balance the positive and negative charges, we look to the least common multiple6: two iron 3+ ions will give 6+, while three 2 oxygen ions will give 6, thereby balancing the overall positive and negative charges. While these names are distinctive and, therefore, are considered chemically-correct, the common system has two significant limitations.What is the empirical formula of aluminium oxide?Īluminium oxide is made out of aluminium and oxygen atoms. The oxygen atom has a 2 charge as an ion. 
For example, the Latin stem for "iron" is "ferr-." Therefore, using the common system, Fe +2 is called the "ferrous ion," and Fe +3 is named as the "ferric ion." The metal cation is named first, followed by the nonmetal anion as illustrated in Figure 5.5.1 5.5. In most cases, the stem, or main portion, of the elemental name used in the common system is typically derived from the element's Latin name. A binary ionic compound is a compound composed of a monatomic metal cation and a monatomic nonmetal anion. 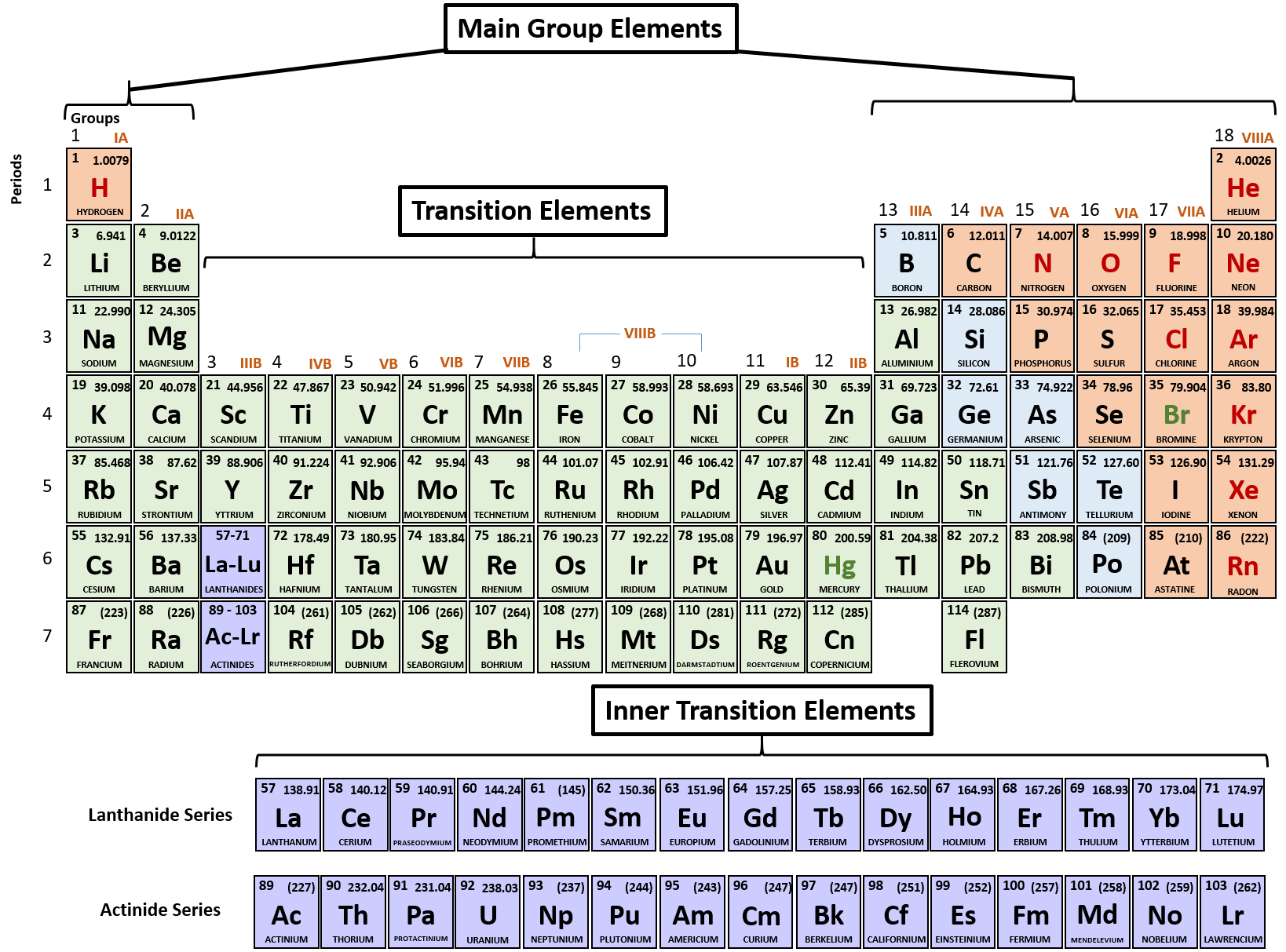
On the periodic table of elements, cobalt is found in period 4 of group 9 and is a part of the d-block. For elements with no stable isotopes, the mass number of the isotope with. 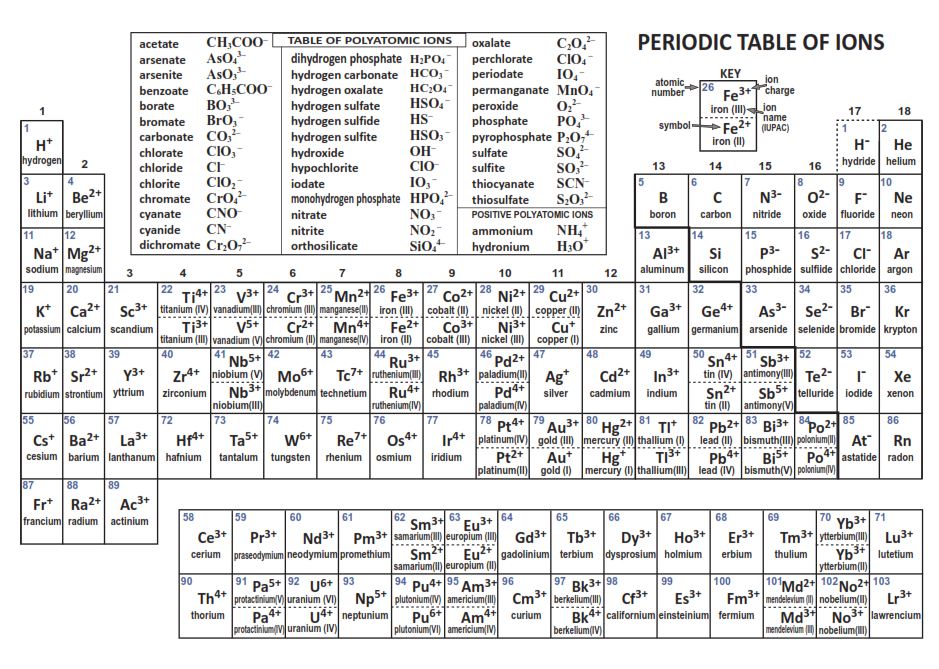
The "-ous" suffix indicates the cation with the lesser relative cation charge, and an "-ic" suffix is used to refer to a cation with a larger charge. Apart from +2, this element commonly forms ions with a +3 charge. 27 Co Cobalt 58.933 28 Ni Nickel 58.693 29 Cu Copper 63.546 30 Zn Zinc 65.38.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
